Decision Support Tool for Evidence-Based Integration of Malaria and Schistosomiasis Control Programs
Research and control programs that strive to combat parasitic diseases often focus on a single infection. These programs have resulted in tremendous amounts of progress towards controlling and reducing the morbidity and mortality rates of diseases like malaria, tuberculosis, and HIV/AIDS, but are not without their shortcomings. Control programs are frequently siloed at the national level and have not addressed other global health concerns when appropriate. For example, these “Big Three” diseases display high levels of co-endemicity with neglected tropical diseases – including demographic and geographic overlap – but these programs are rarely expanded to consider inclusion of concurrent pathogens.
One example of this includes schistosomiasis and malaria control programs. These two diseases co-occur in many regions of Africa and the developing world, predominantly impact children, and have been demonstrated to result in high levels of co-infection at the level of the individual and population. Additionally, schistosomiasis and malaria are known to have complex interactions at the epidemiological and individual level. For example, individuals who are heavily infected with Schistosoma mansoni may have increased malaria susceptibility1, suggesting that the integration of control programs for both diseases could yield significant additive benefits. However, this is rarely the case. Schistosomiasis and malaria control are typically managed under separate programs within Ministries of Health, with separate donors and funding streams, and often with little coordination.
With funding from the Bill & Melinda Gates Foundation, we created a web-based decision support tool to aid policy makers in decisions regarding the benefits integrating malaria and schistosomiasis control programs. With support from collaborators at Talus Analytics, we conducted a thorough literature review, acquired data from collaborators in countries co-endemic for malaria and schistosomiasis, and designed a user interface to translate epidemiological parameters into easily interpretable recommendations. The tool is freely available online.
The tool provides a platform for users to input data regarding the regional age distribution, schistosomiasis control efforts, malaria epidemiology, and malaria control efforts. By incorporating these key inputs, the model provides users with the customized prevalence estimates that support the decision of whether to integrate control programs. Based on this, in order to provide an overall recommendation regarding integrated control and to estimate its potential benefits on malaria prevalence, two model runs are performed. The first “Non-Integrated” simulation represents the “status quo” and is based on the current timing of intervention strategies, as defined by the user. This simulation serves as the reference to which the integrated control program simulation is compared. The second “Integrated” simulation sets the timing of intervention strategies to occur simultaneously.
Based on these simulations, the tool provides recommendations for integration efforts and distribution timelines. The first of these recommendations is based on whether an integrated control program has a meaningful effect (a reduction of five percent or greater) on disease prevalence compared to the non-integrated control programs. The recommended distribution timeline is an intuitive graphic to support the implementation of control programs by public health officials.
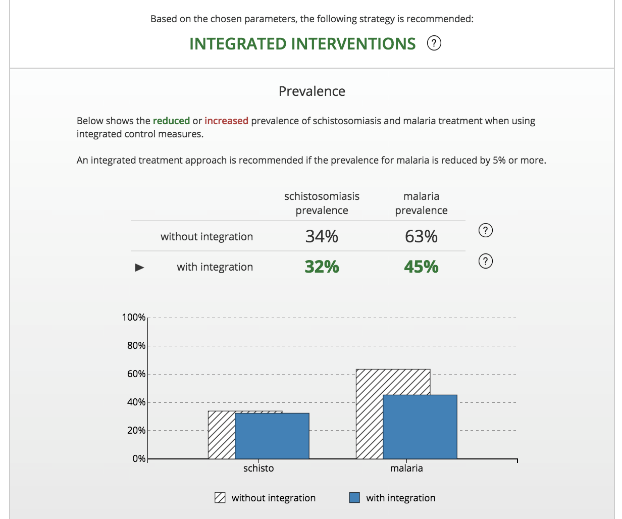
The tool serves as an important proof-of-principle, and represents a step towards achieving more effective and efficient schistosomiasis and malaria control programs. There has been a consensus among end-users in Mali, Uganda, and Yemen that the tool is a useful interface for disease control officers when they are tasked with making decisions about the integration of disease control efforts. Future versions of the tool will strive for improved utility by providing additional detail pertaining to the schistosomiasis portion of the model and by including a cost module that will allow decision makers the opportunity to track estimated expenditures.
Publications
Standley CJ, Graeden E, Kerr J, Sorrell EM, Katz R. Decision support for evidence-based integration of disease control: A proof of concept for malaria and schistosomiasis. PLOS Neglected Tropical Diseases, 2018; 12 (4): e0006328. DOI:10.1371/journal.pntd.0006328
Media Coverage
Acknowledgements: We thank the entire research teams at the Center for Global Health Science and Security and Talus Analytics for their research support in developing the decision support tool. We are also grateful to our colleagues at the Vector Control Division in the Ministry of Health in Uganda, the Malaria Research and Training Center in Mali, and Sana’a University in Yeme


